Applications
We are committed to offering reliable 3D tumor organoid models to advance your preclinical cell and drug development. Our cell models are highly customizable and can be tailored to meet your diverse needs.
Invitrocue’s PDOs are 3D tumor models derived from biopsies or surgical specimens from human. These PDOs exhibit remarkable similarities to the genetics, phenotyping and heterogeneity of the original tumors. These unique features make them an invaluable tool for studying human tumor biology, disease progression and treatment responses.
Conventional in vitro cell lines may not fully represent the complexity of original tumors and lead to poor translatability in clinical settings. This may potentially hinder the development of targeted therapies and personalized medicine in pharmaceutical industry. To overcome this limitation, Invitrocue re-creates the 3D tumor microenvironment by using our proprietary 3D cell culture technology developed in Onco-PDOTM. By accessing our established tumor organoid biobank, we offer to you a series of drug-validated PDOs to support decision-making throughout your preclinical cancer drug and personalized therapeutic strategies.

Fig 1: Organoids for Preclinical Drug Discovery


Breast Cancer
HER2 DAPI

Lung Cancer
CK5 EGFR2 DAPI

Colon Cancer
E-cad P-AKT DAPI

Ovarian Cancer
VIM HER2 PGRMC1 DAPI

We are committed to offering reliable 3D tumor organoid models to advance your preclinical cell and drug development. Our cell models are highly customizable and can be tailored to meet your diverse needs.
To improve the organoid formation rate and effectively retain tumor characteristics.
7 major solid tumors: Breast, Colorectal, Lung, Ovarian, Pancreatic, Gastric, and Nasopharyngeal cancer, and expanding.
Drug responses are clinically proven through IVQ’s Onco-PDOTM test, which has a 75-80% positive predictive accuracy and over 90% negative prediction rate.
Validated with over 75% transplantation success rate from in vitro to in vivo.
PDOs can be expanded in vitro for large-scale drug screening.
Drug resistance and patient treatment history profiles are accessible upon request.


In collaboration with hospitals globally, Invitrocue provides an extensive range of cutting-edge 3D patient-derived organoids (PDO) for academic and industry research. Our proprietary organoids culture technology accurately replicates the genetic, phenotypic and heterogeneous profiles of the original patient tumour. In addition, these organoids have been profiled with Standard of Care (SOC) drugs and validated for in vivo xenograft development. By leveraging this information, Invitrocue offers a ready-to-use, highly translatable and cost-effective in vitro model to accelerate your preclinical drug screening and advance your strategies for personalized cancer treatment.
| S/N | Types of Cancer | Biomarkers | Drug Resistance |
| 1. | Breast Carcinoma |
|
|
| 2. | Colorectal Adenocarcinoma | ||
| 3. | Lung Carcinoma | ||
| 4. | Pancreatic Adenocarcinoma | ||
| 5. | Ovarian Adenocarcinoma | ||
| 6. | Nasopharyngeal Carcinoma | ||
| 7. | Esophageal Carcinoma | ||
| 8. | Bone Sarcoma |
Table 1. Invitrocue’s PDOs, related tumor biomarkers and drug-resistant profile for preclinical in vitro and in vivo services.
Invitrocue is committed to develop innovative products and services. Our high-throughput PDO services enable rapid and accurate preclinical drug efficacy assessments for cancer therapeutics. This section demonstrates case studies that highlight the practical applications and successes of our PDO technology in in drug candidate selection and effective dose-response determination.
| Tumour Info | Details |
| Type | Adenocarcinoma |
| Stage | IV, Metastatic |
| Sampling site | Left paravertebral muscle |
| Biomarkers | EGFR positive (L858R exon 21 mutation) |
| INTEGRATED RESPONSE SCORE (IRS) SUMMARY | |
| DRUGS | IRS SCORE |
| Cisplatin |  |
| Carboplatin |  |
| C1-1 0.2uM |  |
| C1-2 1uM |  |
| C1-3 5uM |  |
| C1-4 10uM |  |
| C1-5 20uM |  |
Figure 2. Rx Score of SOC Drugs and Test Articles on Lung Cancer PDO.
Lung PDO (PDOSG-001-P301) was treated with SOC drugs or test articles at various concentrations in specific 3D lung culture media. Rx score was determined at 72 hours. Increasing doses of test article showed a progressive dose response against Lung PDO and the effect is comparable to SOC drugs like Cisplatin and Carboplatin.
| Tumour Info | Details |
| Type | Pancreatic Cancer |
| Stage | IV, Metastatic |
| Sampling site | Liver |
| INTEGRATED RESPONSE SCORE (IRS) SUMMARY | |
| DRUGS | IRS SCORE |
| Paclitaxel |  |
|
Regorafenib |
No Drug Response |
| Cobimetinib + Everolimus |  |
| Cobimetinib + Panitumumab | 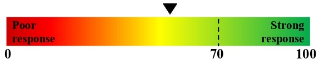 |
| Doxorubicin |  |
| Pemetrexed |  |
| Paclitaxel + Vinorelbine |  |
Figure 3. Rx Score of SOC Drugs on Pancreatic Cancer PDO.
Pancreatic PDO was treated with SOC drugs in specific 3D pancreatic culture media. Rx score was determined at 72 hours. Drug resistance was observed with kinase inhibitor – Regorafenib.
Test article (NDE-001 and NDE-002) responses are represented by Rx.
Rx score indicates the percentage of cell death caused by test article treatments at 72 hours.